Introduction:
Water is the wellspring of life. It is a universal solvent; many types of salts dissolve in water. It is the most important liquid in the world for maintaining the plant and animal life. It fills lakes, streams, the vast oceans, and flows under the ground. Water is a remarkable chemical, an excellent solvent capable of dissolving, to varying degrees, almost anything with which it comes in contact. Water picks up suspended matter as it runs across the ground and absorbs gases from the atmosphere. Impurities in the water come from various sources.
There are different sources of water namely
- Sea Water
- Surface Water
- Ground Water
- Waste Water [Sewage / Industrial]
Common impurities in water:
- Total Dissolved Solids [TDS]: TDS as name signifies is total dissolved salts in water.
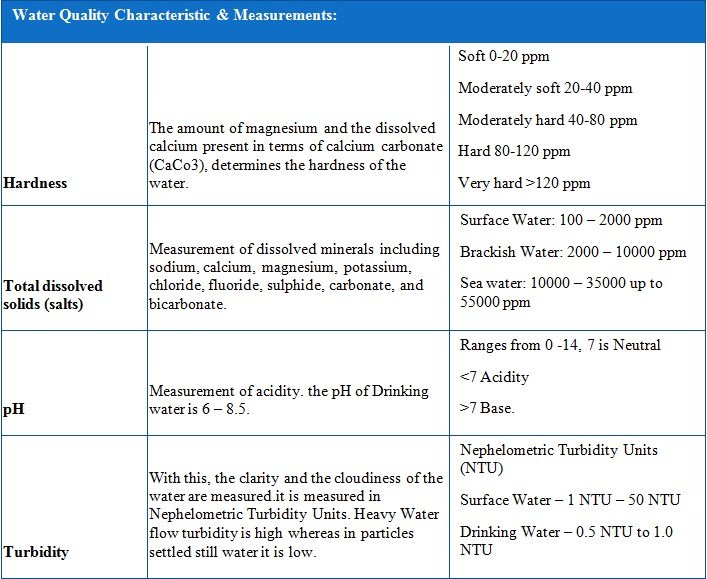
- Hardness: Hardness is the part of TDS which forms hard scales, which composes of calcium Sulfate, Barium Sulfate, Calcium Carbonate, Calcium Fluoride and others like
- Silt
- Organic Impurities
- Biological Impurities
- Metal Oxides etc.
Water Quality:
Rapid industrialization, unplanned resources utilization and poor environmental management have affected the quality of water. Water quality is measured how it is suited for particular purpose for human needsand industrial purposes. With the ever increasing TDS content in raw water and treated effluent RO has been emerged as most economical technology.
Water Quality Index:
To analyze the water quality there are some Water Quality test there are some factors to be considered, they are
 Temperature Temperature |
 Nitrate Nitrate |
 pH pH |
 Phosphate Phosphate |
 Chloride & Salinity Chloride & Salinity |
 Calcium & Water Hardness Calcium & Water Hardness |
 Dissolved Oxygen Dissolved Oxygen |
 Ammonium ion Ammonium ion |
 Turbidity Turbidity |
 TDS TDS |
*water qualities are measured as concentration in milligrams (a thousand of a gram) in one liter of water (mg/L).It can also be
described in “part per million”(ppm) 1 (ppm) = 1 (mg/L).
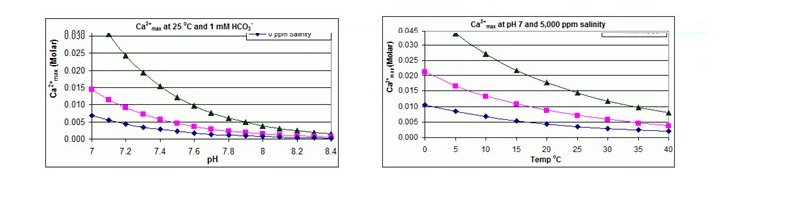
Calcium Carbonate:It is well recognized that calcium carbonates (CaCO3) is one of the main components of scale that is commonly encountered in chemical and related industries. The calcium carbonate scale often grows extensively, causing major operational difficulties.The temperature and pH values affect the kinetics of crystal formation.
The solubility of Calcium Carbonate is typically limited to a LSI (Langlier Saturation Index) value of positive 1.8 to 2.5.Calcium Carbonate solubility is measured using LSI (Langlier Saturation Index) forbrackish waters or SDSI (Stiff and Davis Saturation Index) for seawaters and is lower with increasing temperature and increasing pH.
The solubility product for CaCO3 and the dissociation constants for the DIC (Dissolved Inorganic Carbon) species are all substantially affected by temperature and salinity with the overall effect that Ca2+max increases from fresh to salt water, and decreases with rising temperature, pH, or added bicarbonate level, as illustrated in the accompanying graphs.



To calculate SDI follow these steps:
Step 1: Measure the time required to filter a fixed volume of water eg:- 500ml through a standard
0.45μm pore size microfiltration membrane at a constant pressure of 30 psi (2.07 bar). Record this as
Ti, or T initial.
Step 2: Take additional time measurements, normally after 5, 10 and 15 minutes (after silt build up)
Step 3: Calculate the Plugging Factor after 5, 10 and 15 minutes as follows:
PF5 = (1-Ti /T5)*100
PF10 = (1-Ti /T10)*100
PF15 = (1-Ti /T15)*100
Step 4: The SDI value is then determined at each interval as SDI = PF/T.


